CARTIA XT® brand of DILTIAZEM HYDROCHLORIDE EXTENDED-RELEASE CAPSULES, USP(ONCE-A-DAY DOSAGE)Rx only
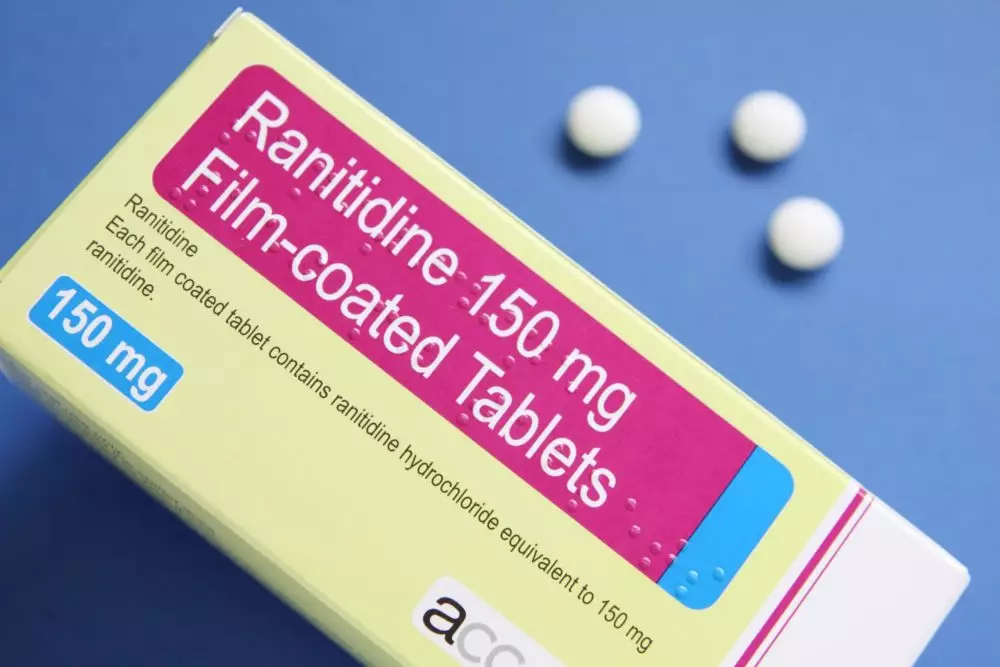
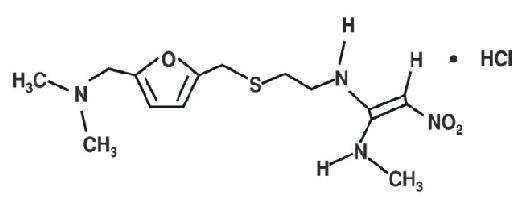
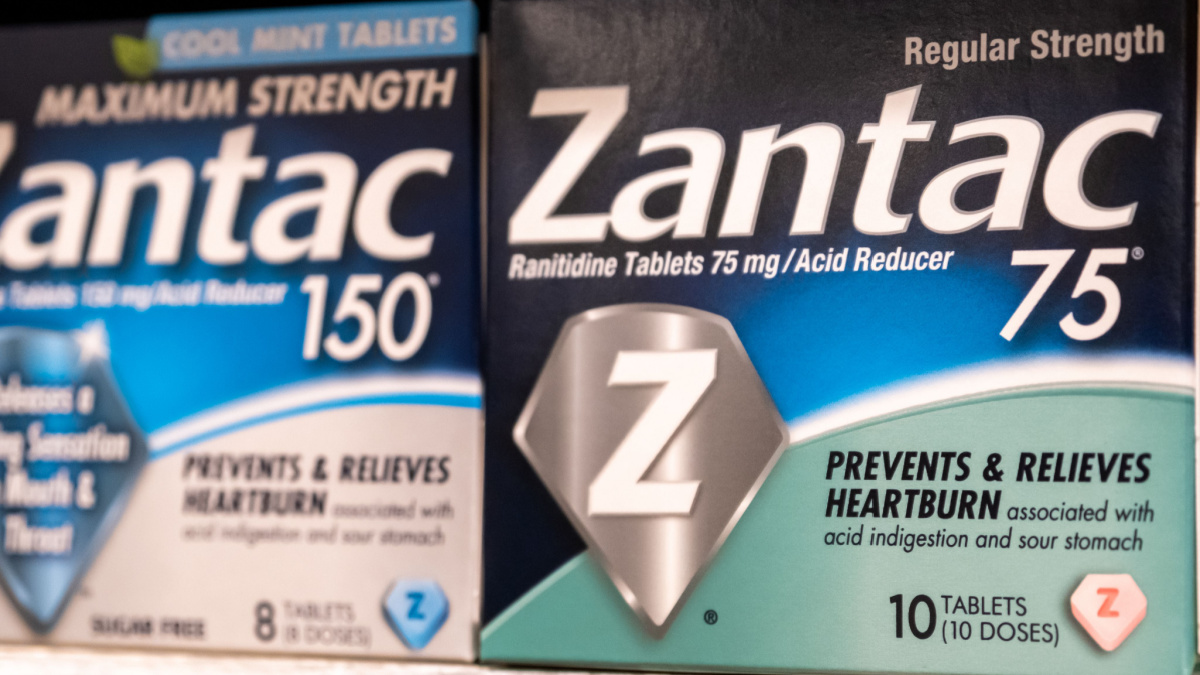
Information Update - Additional ranitidine products recalled, including Zantac; request to stop distribution remains in place while Health Canada continues to assess NDMA