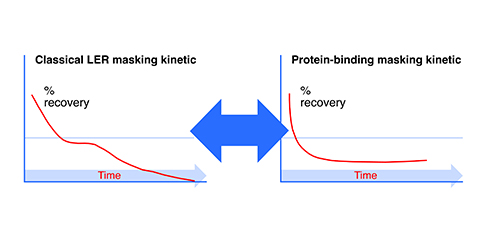
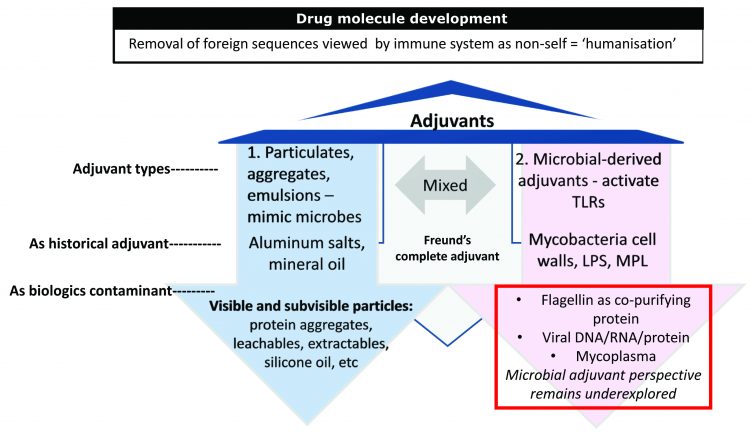
The Truth of Endotoxin Values - Points for Consideration During Investigation of Aberrant BET Results | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology
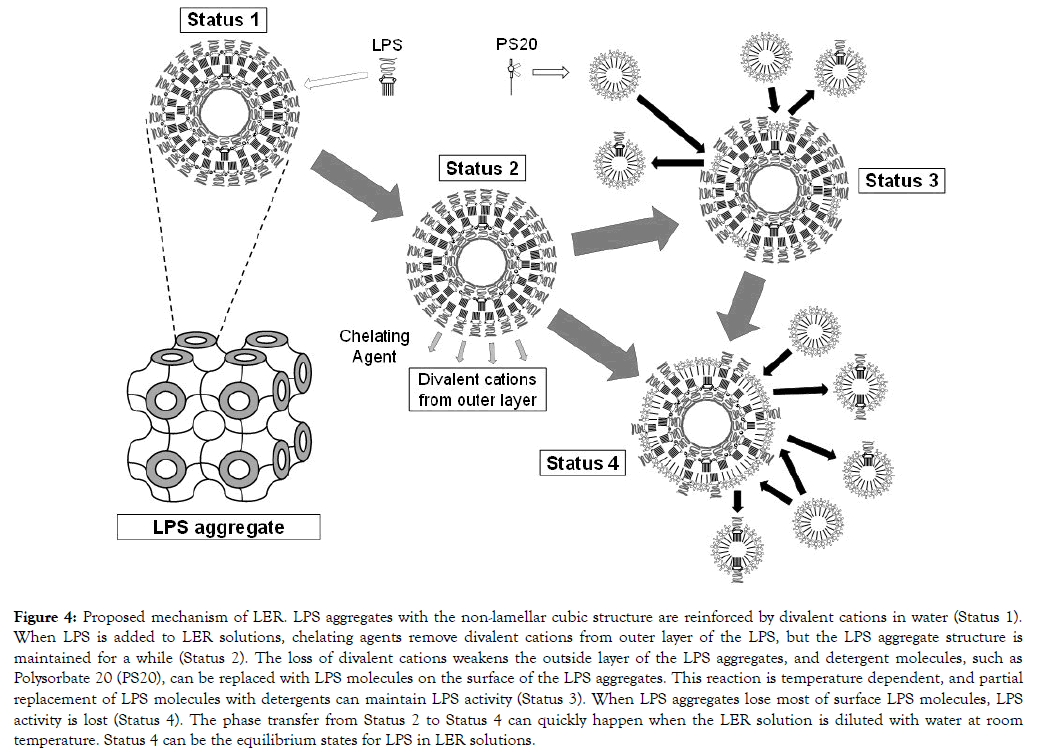
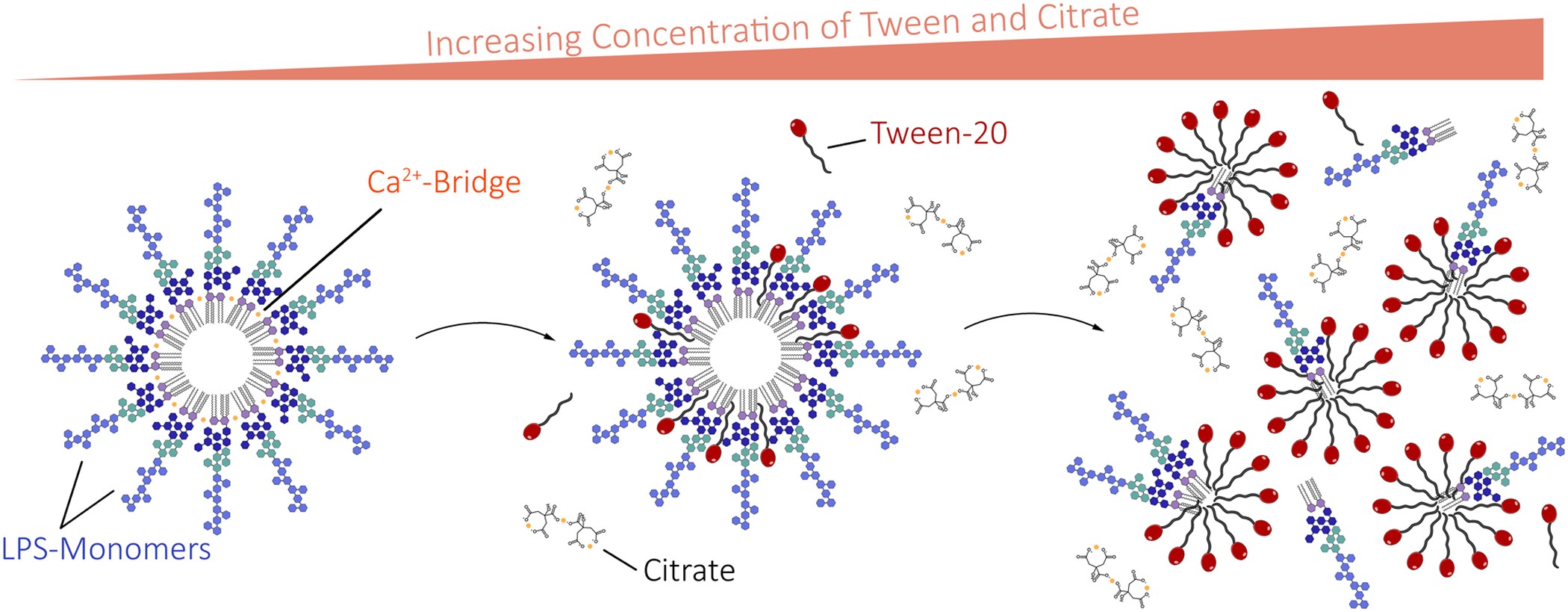
Mechanism of Low Endotoxin Recovery Caused by a Solution Containing a Chelating Agent and a Detergent

Evidence Against a Bacterial Endotoxin Masking Effect in Biologic Drug Products by Limulus Amebocyte Lysate Detection | PDA Journal of Pharmaceutical Science and Technology

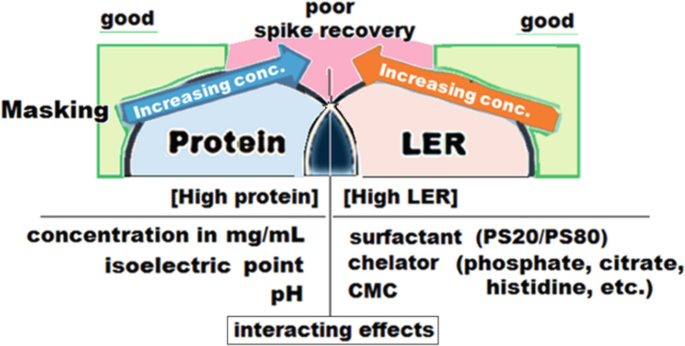
Low Endotoxin Recovery (LER) is today one of authorities serious concerns regarding pyrogen testing - A3P - Pharmaceutical & Biotechnology Industry

Masking of an endogenous endotoxin contamination. Endotoxin activity of... | Download Scientific Diagram
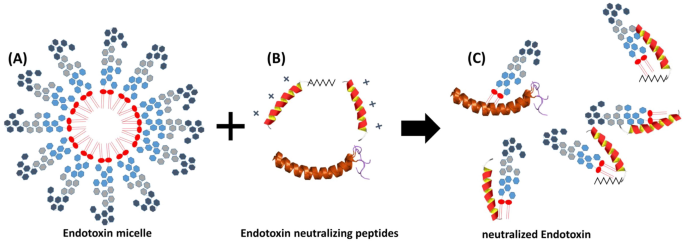
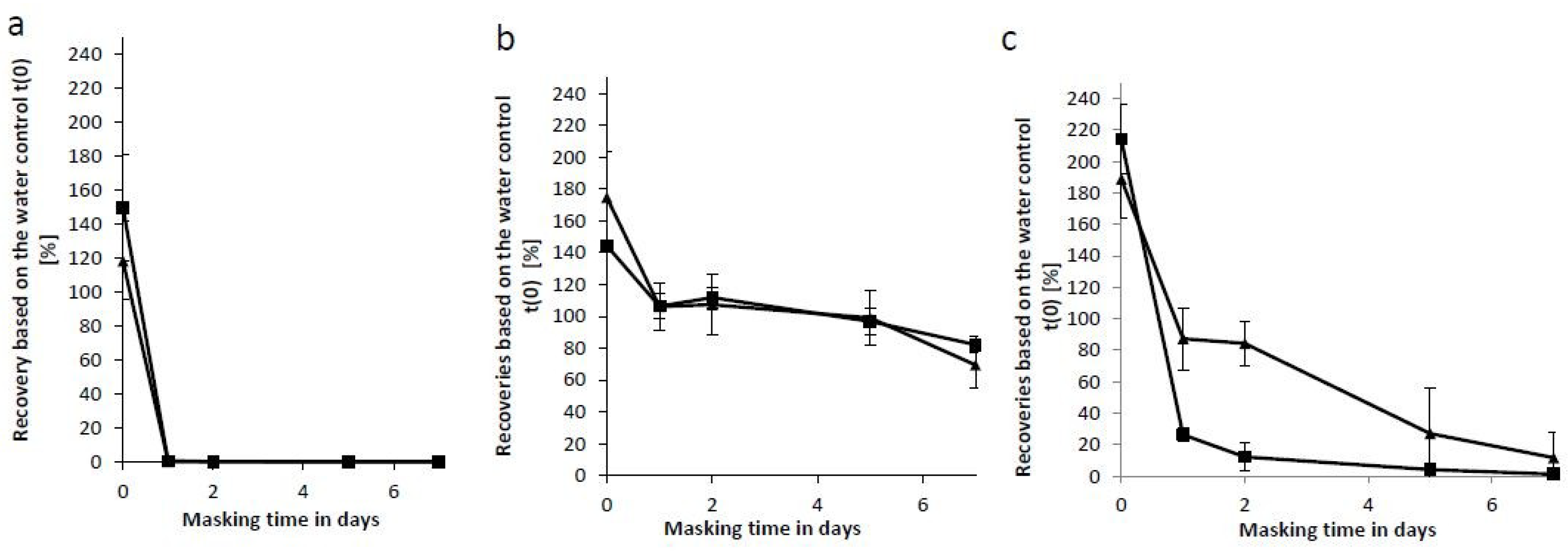
An in vitro study on factors affecting endotoxin neutralization in human plasma using the Limulus amebocyte lysate test | Scientific Reports

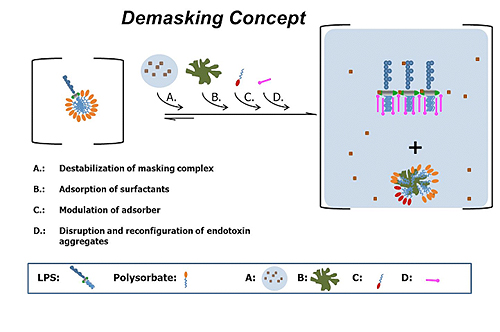
Bacterial Endotoxin Testing: New Method for Unmasking Endotoxin in BiopharmaceuticalsBioProcess International

Current technologies to endotoxin detection and removal for biopharmaceutical purification - Schneier - 2020 - Biotechnology and Bioengineering - Wiley Online Library

Fit For Purpose” Endotoxin Analytes | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

Bacterial Endotoxin Testing: New Method for Unmasking Endotoxin in BiopharmaceuticalsBioProcess International